CSET Requirement 2.3c: Know the location of the ozone layer in the upper atmosphere, explain its role in absorbing ultraviolet radiation, and explain the way in which this layer varies both naturally and in response to human activities.
The Ozone Layer

Ozone is a gas that occurs naturally in our atmosphere. Most of it is concentrated in the ozone layer, a region located in the stratosphere several miles above the surface of the Earth. Although ozone represents only a small fraction of the gas present in the atmosphere, it plays a vital role by shielding humans and other life from harmful ultraviolet light from the Sun. Human activities in the last several decades have produced chemicals, such as chlorofluorocarbons (CFCs), which have been released into the atmosphere and have contributed to the depletion of this important protective layer. When scientists realized the destructive effect these chemicals could have on the ozone layer, international agreements were put in place to limit such emissions. As a result, it is expected that the ozone layer will recover in the coming decades.
Ozone is also a greenhouse gas in the upper atmosphere and, therefore, plays a role in Earth's climate. The increases in primary greenhouse gases, such as carbon dioxide, may affect how the ozone layer recovers in coming years. Understanding precisely how ozone abundances will change in a future with diminished chlorofluorocarbon emissions and increased emissions of greenhouse gases remains an important challenge for atmospheric scientists.
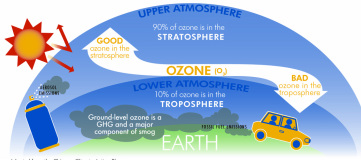
Origins and UV RadiationOzone in the Earth's stratosphere is created by ultraviolet light striking oxygen molecules containing two oxygen atoms(O2), splitting them into individual oxygen atoms (atomic oxygen); the atomic oxygen then combines with unbroken O2 to create ozone, O3. The ozone molecule is also unstable (although, in the stratosphere, long-lived) and when ultraviolet light hits ozone it splits into a molecule of O2 and an atom of atomic oxygen, a continuing process called the ozone-oxygen cycle, thus creating an ozone layer in the stratosphere, the region from about 10 to 50 kilometres (33,000 to 160,000 ft) above Earth's surface. About 90% of the ozone in our atmosphere is contained in the stratosphere.
Although the concentration of the ozone in the ozone layer is very small, it is vitally important to life because it absorbs biologically harmful ultraviolet (UV) radiation coming from the sun. UV-C, which would be very harmful to all living things, is entirely screened out by a combination of dioxygen and ozone by around 35 kilometres (115,000 ft) altitude. UV-B radiation can be harmful to the skin and is the main cause of sunburn; excessive exposure can also cause genetic damage, resulting in problems such as skin cancer. The ozone layer is very effective at screening out UV-B; for radiation, the intensity at the top of the atmosphere is 350 million times stronger than at the Earth's surface. Nevertheless, some UV-B, particularly at its longest wavelengths, reaches the surface.
Ozone is also a greenhouse gas in the upper atmosphere and, therefore, plays a role in Earth's climate. The increases in primary greenhouse gases, such as carbon dioxide, may affect how the ozone layer recovers in coming years. Understanding precisely how ozone abundances will change in a future with diminished chlorofluorocarbon emissions and increased emissions of greenhouse gases remains an important challenge for atmospheric scientists.
Origins and UV RadiationOzone in the Earth's stratosphere is created by ultraviolet light striking oxygen molecules containing two oxygen atoms(O2), splitting them into individual oxygen atoms (atomic oxygen); the atomic oxygen then combines with unbroken O2 to create ozone, O3. The ozone molecule is also unstable (although, in the stratosphere, long-lived) and when ultraviolet light hits ozone it splits into a molecule of O2 and an atom of atomic oxygen, a continuing process called the ozone-oxygen cycle, thus creating an ozone layer in the stratosphere, the region from about 10 to 50 kilometres (33,000 to 160,000 ft) above Earth's surface. About 90% of the ozone in our atmosphere is contained in the stratosphere.
Although the concentration of the ozone in the ozone layer is very small, it is vitally important to life because it absorbs biologically harmful ultraviolet (UV) radiation coming from the sun. UV-C, which would be very harmful to all living things, is entirely screened out by a combination of dioxygen and ozone by around 35 kilometres (115,000 ft) altitude. UV-B radiation can be harmful to the skin and is the main cause of sunburn; excessive exposure can also cause genetic damage, resulting in problems such as skin cancer. The ozone layer is very effective at screening out UV-B; for radiation, the intensity at the top of the atmosphere is 350 million times stronger than at the Earth's surface. Nevertheless, some UV-B, particularly at its longest wavelengths, reaches the surface.
Natural distribution of the Ozone Layer

Total ozone varies strongly withv latitude over the globe, with the largest values occurring at middle and high latitudes during all seasons (see picture). This is the result of ozone production rates from solar ultraviolet radiation that are highest on average in the tropics, and the large-scale air circulation in the stratosphere that slowly transports tropical ozone toward the poles. Ozone accumulates at middle and high latitudes, increasing the thickness (or vertical extent) of the ozone layer and, at the same time, total ozone. In contrast, the values of total ozone are the lowest in the tropics in all seasons (except in the ozone hole) because the thickness of the ozone layer is smallest there.
Total ozone also varies with seasons. Ozone shows a maximum at high latitudes during spring as a result of increased transport of ozone from its source region in the tropics toward the polar regions during late fall and winter. This ozone transport is much weaker during the summer and early fall periods and is weaker overall in the Southern Hemisphere. An important feature of seasonal ozone changes is the natural chemical destruction that occurs when daylight is continuous in the summer polar stratosphere, which causes total ozone to decrease gradually. This natural seasonal cycle can be observed clearly in the Northern Hemisphere with increasing values in Arctic total ozone during winter, a clear maximum in spring, and decreasing values from summer to fall. In the Antarctic, however, a pronounced minimum in total ozone is observed during spring. The minimum is a consequence of the “ozone hole,” which describes the widespread chemical destruction of ozone by ozone-depleting substances.In the late 1970s, before the ozone hole appeared each year, much higher ozone values were found in Antarctic spring. Now, the lowest values of total ozone across the globe and all seasons are found every spring in the Antarctic. After spring, these low values disappear from total ozone maps as polar air mixes with lower-latitude air containing much higher ozone values. In the tropics, the total ozone changes through the spring-summer-fall-winter progression of the seasons are much smaller than in the polar regions. This is because seasonal changes in both sunlight and ozone transport are smaller in the tropics than in the polar regions.
Total ozone varies strongly with latitude and longitude within seasons. These patterns, which change on daily to weekly timescales, come about for two reasons. First, natural air motions mix and blend air between regions of the stratosphere that have high ozone values and those that have low ozone values. Tropospheric weather systems can temporarily change the thickness of the ozone layer in a region, and thereby change total ozone. The geographical variation in these air motions in turn causes variations in the distribution of total ozone. Second, ozone variations occur as a result of changes in the balance of chemical production and loss processes as air moves to and from different locations over the globe. This balance, for example, is very sensitive to the amount of sunlight in a region.
Total ozone also varies with seasons. Ozone shows a maximum at high latitudes during spring as a result of increased transport of ozone from its source region in the tropics toward the polar regions during late fall and winter. This ozone transport is much weaker during the summer and early fall periods and is weaker overall in the Southern Hemisphere. An important feature of seasonal ozone changes is the natural chemical destruction that occurs when daylight is continuous in the summer polar stratosphere, which causes total ozone to decrease gradually. This natural seasonal cycle can be observed clearly in the Northern Hemisphere with increasing values in Arctic total ozone during winter, a clear maximum in spring, and decreasing values from summer to fall. In the Antarctic, however, a pronounced minimum in total ozone is observed during spring. The minimum is a consequence of the “ozone hole,” which describes the widespread chemical destruction of ozone by ozone-depleting substances.In the late 1970s, before the ozone hole appeared each year, much higher ozone values were found in Antarctic spring. Now, the lowest values of total ozone across the globe and all seasons are found every spring in the Antarctic. After spring, these low values disappear from total ozone maps as polar air mixes with lower-latitude air containing much higher ozone values. In the tropics, the total ozone changes through the spring-summer-fall-winter progression of the seasons are much smaller than in the polar regions. This is because seasonal changes in both sunlight and ozone transport are smaller in the tropics than in the polar regions.
Total ozone varies strongly with latitude and longitude within seasons. These patterns, which change on daily to weekly timescales, come about for two reasons. First, natural air motions mix and blend air between regions of the stratosphere that have high ozone values and those that have low ozone values. Tropospheric weather systems can temporarily change the thickness of the ozone layer in a region, and thereby change total ozone. The geographical variation in these air motions in turn causes variations in the distribution of total ozone. Second, ozone variations occur as a result of changes in the balance of chemical production and loss processes as air moves to and from different locations over the globe. This balance, for example, is very sensitive to the amount of sunlight in a region.
Ozone response due to human activities
The ozone layer is deteriorating due to the release of pollution containing the chemicals chlorine and bromine. Chlorofluorocarbons (CFCs), chemicals found mainly in spray aerosols heavily used by industrialized nations for much of the past 50 years, are the primary culprits in ozone layer breakdown. When CFCs reach the upper atmosphere, they are exposed to ultraviolet rays, which causes them to break down into substances that include chlorine. The chlorine reacts with the oxygen atoms in ozone and rips apart the ozone molecule. One atom of chlorine can destroy more than a hundred thousand ozone molecules, according to the the U.S. Environmental Protection Agency. The ozone layer above the Antarctic has been particularly impacted by pollution since the mid-1980s. This region’s low temperatures speed up the conversion of CFCs to chlorine. In the southern spring and summer, when the sun shines for long periods of the day, chlorine reacts with ultraviolet rays, destroying ozone on a massive scale, up to 65 percent. In other regions, the ozone layer has deteriorated by about 20 percent. About 90 percent of CFCs currently in the atmosphere were emitted by industrialized countries in the Northern Hemisphere, including the United States and Europe. These countries banned CFCs by 1996, and the amount of chlorine in the atmosphere is falling now. But scientists estimate it will take another 50 years for chlorine levels to return to their natural levels.
