CSET Requirement 2.2a: Describe the chemical and physical properties of sea water.
The most important components of seawater that influence life forms are salinity, temperature, dissolved gases (mostly oxygen and carbon dioxide), nutrients, and pH.
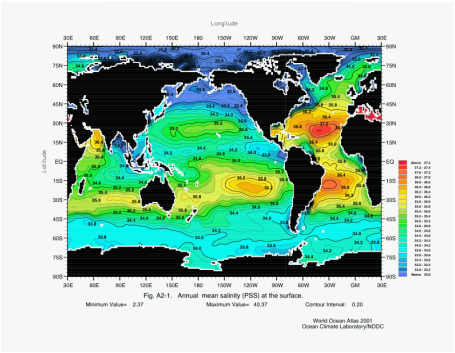
Physical Property: SALINITY
About 70% of Earth is covered with water and 97% of that amount is seawater. Water contains about 3.5% by weight of salt, this means that one kilogram (1,000 grams) will have 35 grams of dissolved salts. This measurement is also known as 35 parts per thousand or 35 ppt. Salinity varies and its combination with temperature has important effects on ocean currents. (Please research arctic melt and its effect on the North Atlantic/Gulf Stream currents) The most common factor is the relative amount of evaporation or precipitation in an area. If there is more evaporation than precipitation then the salinity increases (since salt is not evaporated into the atmosphere). If there is more precipitation (rain) than evaporation then the salinity decreases. Another factor that can change the salinity in the ocean is due to a very large river emptying into the ocean. The runoff from most small streams and rivers is quickly mixed with ocean water by the currents and has little effect on salinity. But large rivers (like the Amazon River in South America) may make the ocean have little or no salt content for over a mile or more out to sea. The freezing and thawing of ice also affects salinity. The thawing of large icebergs (made of frozen fresh water and lacking any salt) will decrease the salinity while the actual freezing of seawater will increase the salinity temporarily. This temporary increase happens in the first stages of the freezing of seawater when small ice crystals form at about minus 2 degrees Centigrade. These tiny, needle-like ice crystals are frozen freshwater and the salts are not part of them so the liquid between these crystals becomes increasingly salty to the point of it being a brine. Eventually though, as seawater freezes, the ice crystals trap areas with brine and the entire large piece of frozen seawater (ice floe) is salty.
About 70% of Earth is covered with water and 97% of that amount is seawater. Water contains about 3.5% by weight of salt, this means that one kilogram (1,000 grams) will have 35 grams of dissolved salts. This measurement is also known as 35 parts per thousand or 35 ppt. Salinity varies and its combination with temperature has important effects on ocean currents. (Please research arctic melt and its effect on the North Atlantic/Gulf Stream currents) The most common factor is the relative amount of evaporation or precipitation in an area. If there is more evaporation than precipitation then the salinity increases (since salt is not evaporated into the atmosphere). If there is more precipitation (rain) than evaporation then the salinity decreases. Another factor that can change the salinity in the ocean is due to a very large river emptying into the ocean. The runoff from most small streams and rivers is quickly mixed with ocean water by the currents and has little effect on salinity. But large rivers (like the Amazon River in South America) may make the ocean have little or no salt content for over a mile or more out to sea. The freezing and thawing of ice also affects salinity. The thawing of large icebergs (made of frozen fresh water and lacking any salt) will decrease the salinity while the actual freezing of seawater will increase the salinity temporarily. This temporary increase happens in the first stages of the freezing of seawater when small ice crystals form at about minus 2 degrees Centigrade. These tiny, needle-like ice crystals are frozen freshwater and the salts are not part of them so the liquid between these crystals becomes increasingly salty to the point of it being a brine. Eventually though, as seawater freezes, the ice crystals trap areas with brine and the entire large piece of frozen seawater (ice floe) is salty.
Physical Property: TEMPERATURE and DENSITY

Temperature and salinity share an inverse relationship. The freezing point of seawater decreases as salinity increases. At normal salinity, freezing point is at 28F. Temperature and density also share an inverse relationship. As temperature increases, the space between water molecules—also known as density, decreases. The cooler seawater is, the denser it becomes. Normal seawater density is at 1,020 kg per cubic meter, cooler waters can reach up to 1,050. Increasing salinity also increases density. Given two layers of seawater with similar salinity, the cooler layer will be denser and fall below the warmer layer when temperature is taken into account. There is more to add to your confusion though! Temperature has a greater effect on the density of water than salinity does. So a layer of water with higher salinity can actual float on top of water with lower salinity if the layer with higher salinity is warmer than the lower salinity layer.
The temperature of the ocean decreases and decreases as you go to the bottom of the ocean. So, the density of ocean water increases and increases as you go to the bottom of the ocean. The deep ocean is layered with the densest water on bottom and the lightest water on top. Circulation in the depths of the ocean is horizontal making water move along the layers with the same density.
The temperature of seawater varies with the amount of sun that hits that area. This includes the length of time as well as the angle of the sun's rays. The longer the time and the more direct the rays of the sun fall on the ocean, the greater the temperature of seawater. Thus, tropical areas that get more year-round sun and more direct sun (almost 90 degrees, straight down for most of the year at noon) have warmer surface waters than polar areas that may have no sun at all for several months each year and then very steep angles of the sun's rays (never directly overhead at noon). Knowing this about ocean water helps us understand that surface ocean temperatures are warm in the tropics and cooler at the poles.
Currents also have a lot to do with temperature and density in sea water. Ocean water is constantly churning underneath. The difference in density of cold water versus density of warmer water is responsible for ocean currents and upwelling. Warm seawater floats and cold dense seawater sinks, so ocean temperatures also vary across the surface and into the depths.
Case Study: The California Current is a Pacific Ocean current that runs alongside the state of California. It begins off the southern end of British Columbia and finishes off southern Baja California. This movement of northern waters southward makes the coastal waters cooler. Additionally, upwelling of cooler deeper waters occur as winds push surface waters away from the shore allowing cooler waters to replace them, this lowers the temperature of the already cool California coastal waters. This translates into cold coastal waters during the summer, stretching from Oregon to Baja California. (This does not include the coastal water surrounding San Diego. There is a warm water anomaly off San Diego). Fog forms along the California coast as air in coastal valleys (Bay area or central valleys) heat up and expand during the day. This air rises, is displaced and replaced by air drawn in from offshore. A narrower weaker current flows south to north in the winter but by late February disappears as the California Current overrides it. By late spring and early summer, the California Current has fully arrived to southern California where a phenomena arises- June Gloom. When the ocean is that cold, water vapor in the air cools down to form fog or clouds when the dew point is reached (the temperature at which water vapor in the air starts to condense). (I decided to talk about this here because it shows up on the test.)
The temperature of the ocean decreases and decreases as you go to the bottom of the ocean. So, the density of ocean water increases and increases as you go to the bottom of the ocean. The deep ocean is layered with the densest water on bottom and the lightest water on top. Circulation in the depths of the ocean is horizontal making water move along the layers with the same density.
The temperature of seawater varies with the amount of sun that hits that area. This includes the length of time as well as the angle of the sun's rays. The longer the time and the more direct the rays of the sun fall on the ocean, the greater the temperature of seawater. Thus, tropical areas that get more year-round sun and more direct sun (almost 90 degrees, straight down for most of the year at noon) have warmer surface waters than polar areas that may have no sun at all for several months each year and then very steep angles of the sun's rays (never directly overhead at noon). Knowing this about ocean water helps us understand that surface ocean temperatures are warm in the tropics and cooler at the poles.
Currents also have a lot to do with temperature and density in sea water. Ocean water is constantly churning underneath. The difference in density of cold water versus density of warmer water is responsible for ocean currents and upwelling. Warm seawater floats and cold dense seawater sinks, so ocean temperatures also vary across the surface and into the depths.
Case Study: The California Current is a Pacific Ocean current that runs alongside the state of California. It begins off the southern end of British Columbia and finishes off southern Baja California. This movement of northern waters southward makes the coastal waters cooler. Additionally, upwelling of cooler deeper waters occur as winds push surface waters away from the shore allowing cooler waters to replace them, this lowers the temperature of the already cool California coastal waters. This translates into cold coastal waters during the summer, stretching from Oregon to Baja California. (This does not include the coastal water surrounding San Diego. There is a warm water anomaly off San Diego). Fog forms along the California coast as air in coastal valleys (Bay area or central valleys) heat up and expand during the day. This air rises, is displaced and replaced by air drawn in from offshore. A narrower weaker current flows south to north in the winter but by late February disappears as the California Current overrides it. By late spring and early summer, the California Current has fully arrived to southern California where a phenomena arises- June Gloom. When the ocean is that cold, water vapor in the air cools down to form fog or clouds when the dew point is reached (the temperature at which water vapor in the air starts to condense). (I decided to talk about this here because it shows up on the test.)
Chemical Properties of Seawater

Seawater is essentially water and a complex solution of salts.These salts comprise approximately 3.5% (by weight) of seawater.This percentage may appear small but the quantities of salt in the ocean are enormous.If all the ocean water was evaporated, there would still be a layer of salt 60m thick covering the entire ocean floor.Several elements can be found in this complex solution of salts in seawater. The most abundant salt is sodium chloride, otherwise known as common table salt. Sodium chloride comprises almost 70% of all the elements in the salt solution.Sodium chloride combined with the next three most abundant salts: magnesium chloride, sodium sulfate and calcium chloride comprise about 99% of the elements found in seawater. .
six elements and compounds comprise sea salts: chlorine [55% (Cl-)], sodium[30.6% (Na+)], sulfur [7.7% (SO4-2)], magnesium [3.65% (Mg+2)], calcium [1.17% (Ca+2)], and potassium [1.13% (K+)].
Trace elements make up the remainder of composition which include chromium, cobalt, uranium, fluoride, iodine, manganese, selenium, and zinc to name a few.
Test Sample Question: The soils of some areas of the westernmost San Joaquin Valley have high levels of naturally occurring selenium, a trace mineral that can be toxic at high concentrations. Which of the following is the major source of the selenium found in these soils?A. dust that settles along the western B. underlying bedrock strata derived C. river sediments washing down from D. drought-tolerant plants
from ocean sediments side of the San Joaquin Valley the Coast Range that concentrate trace minerals
six elements and compounds comprise sea salts: chlorine [55% (Cl-)], sodium[30.6% (Na+)], sulfur [7.7% (SO4-2)], magnesium [3.65% (Mg+2)], calcium [1.17% (Ca+2)], and potassium [1.13% (K+)].
Trace elements make up the remainder of composition which include chromium, cobalt, uranium, fluoride, iodine, manganese, selenium, and zinc to name a few.
Test Sample Question: The soils of some areas of the westernmost San Joaquin Valley have high levels of naturally occurring selenium, a trace mineral that can be toxic at high concentrations. Which of the following is the major source of the selenium found in these soils?A. dust that settles along the western B. underlying bedrock strata derived C. river sediments washing down from D. drought-tolerant plants
from ocean sediments side of the San Joaquin Valley the Coast Range that concentrate trace minerals
DISSOLVED GASES

Seawater also contains small amounts of dissolved gases. Many of these gases are added to seawater from the atmosphere through the constant stirring of the sea surface by wind and waves. The concentration of gases that can be dissolved into seawater from the atmosphere is determined by temperature and salinity of the water. Increasing the temperature or salinity reduces the amount of gas that ocean water can dissolve. Some of the important atmospheric gases found in seawater include: nitrogen, oxygen, carbon dioxide (in the form of bicarbonate HCO3), argon, helium, and neon. Compared to the other atmospheric gases, the amount of carbon dioxide dissolved in saturated seawater is unusually large.
Some gases found within seawater are also involved in oceanic organic and inorganic processes that are indirectly related to the atmosphere. For example, oxygen and carbon dioxide may be temporally generated or depleted by such processes to varying concentrations at specific locations within the ocean.
Some gases found within seawater are also involved in oceanic organic and inorganic processes that are indirectly related to the atmosphere. For example, oxygen and carbon dioxide may be temporally generated or depleted by such processes to varying concentrations at specific locations within the ocean.
